|
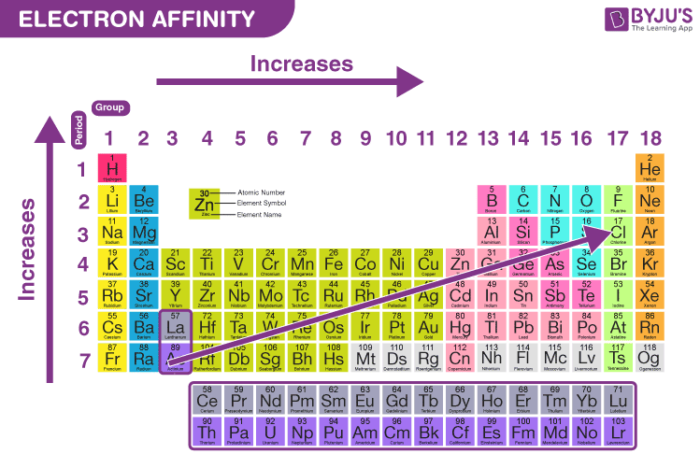
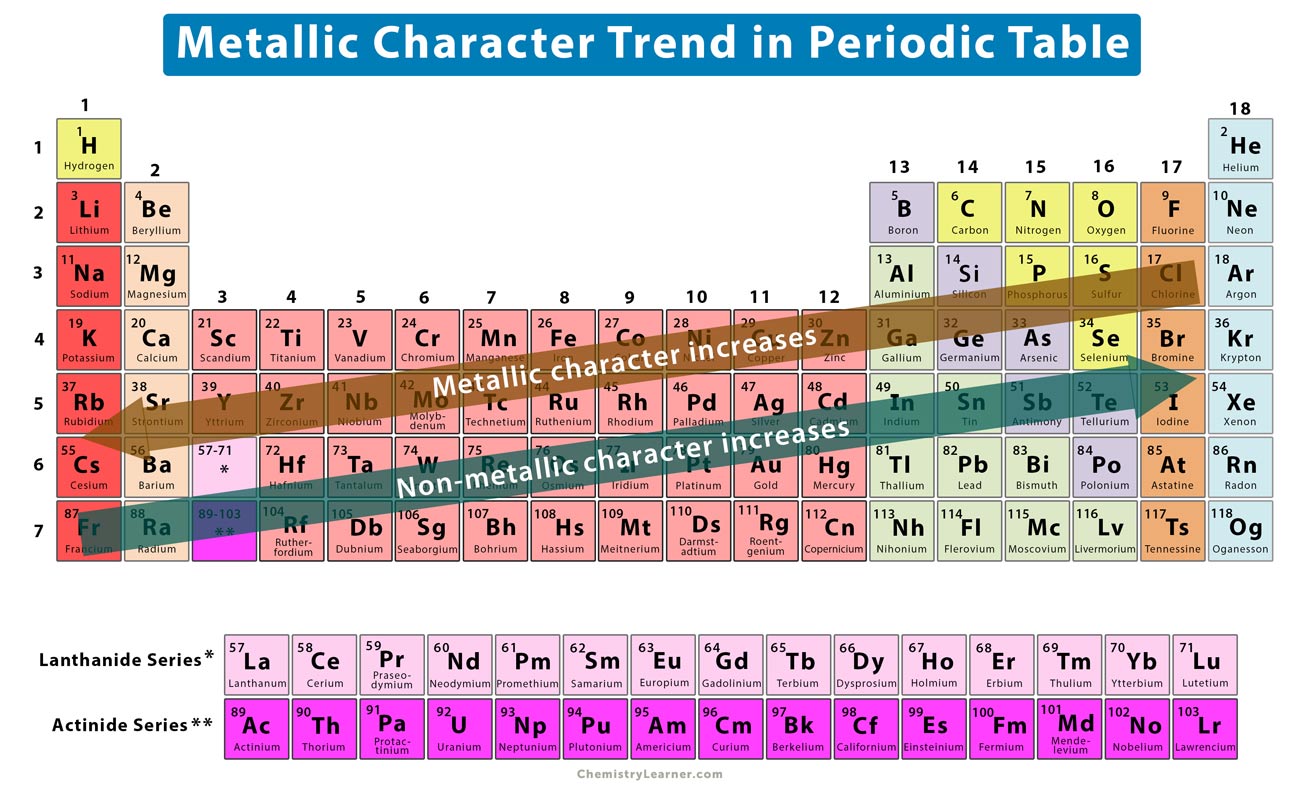
The melting point is the temperature at which a solid begins to melt at a particular pressure. State the trends of the following across the periods and down the groups Also, the larger the charge on a cation, the smaller the ionic radius and the larger the charge on the anion, the larger the ionic radius. Note: Ionic radius generally increases down the groups and decreases across the periods.Ĭations formed by electron loss are smaller in size than the neutral atom from which they are formed and anions formed by electron gain are larger in size than the neutral atoms that form them. Ionic radius decreases for metal across the periods and increases for non – metals.Electron affinity decreases from left across each period while it increases down the groups.Electropositivity decreases from left to right across the period while it increases down the groups from top to bottom.Electronegativity increases from left to right across each period while it decreases down the groups from top to bottom.Ionization energy increases across the periods left to right while it decreases down the groups from top to the bottom.Atomic radius decreases across the periods from left to right while it increases down the groups from top to bottom.Define two of the stated atomic physical properties.State five atomic physical properties of elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
